Over the last few centuries, our understanding of atomic structure has evolved significantly.
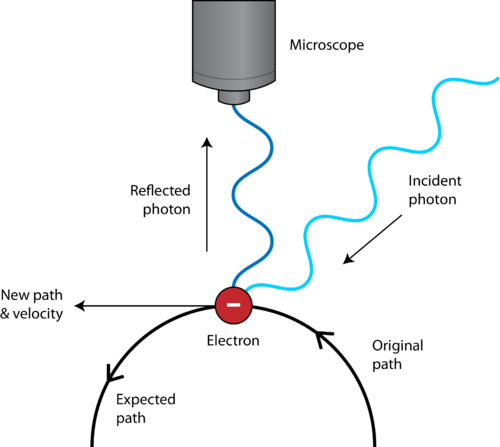
The evolution of our understanding of atomic structure Schrödinger also formulated the Schrodinger-wave equations to accurately calculate electron energy levels. In this model, electrons do not revolve around the nucleus in fixed orbits but exist as electron clouds in atomic orbitals. Erwin Schrödinger developed the quantum mechanical model of an atom, based on wave-particle duality and the uncertainty principle. Quantum mechanics and the Schrodinger-wave equationsīohr's model, although widely accepted, did not satisfy the Heisenberg Uncertainty Principle, which states that it is impossible to know an electron's exact position and trajectory simultaneously. An electron falling to a lower energy orbit would emit radiation, thus maintaining atomic stability.

To address this issue, Bohr incorporated the concept of quantized energy levels and proposed that electrons moved in fixed orbits or shells. According to classical physics, electrons orbiting a positively charged center would lose energy and collapse into the nucleus, making atoms unstable. However, Bohr found a discrepancy in the model regarding the stability of atoms. Rutherford's atomic theory prompted Niels Bohr to investigate further. Based on his observations, Rutherford proposed a new atomic model in which the atom's mass was concentrated in a positively charged nucleus, with electrons orbiting around it like planets around the sun. By bombarding a thin sheet of gold foil with alpha particles, Rutherford observed that some particles were deflected at angles greater than 90 degrees. Not convinced by Thompson's model, Ernest Rutherford conducted the gold-foil experiment. Ernest Rutherford and the gold-foil experiment Through these experiments, Thompson developed the "plum pudding" model, describing atoms as spheres of positive charge with negatively charged particles embedded in them. His experiments involved applying high voltage to metal electrodes, which produced a stream of particles called cathode rays. Thompson challenged the concept of indivisible particles by conducting experiments with cathode ray tubes. Thompson and the plum pudding modelĮnglish physicist J.J. Dalton proposed that everything in the world was made up of atoms that were unique to each element. He discovered that gases reacted with each other in fixed ratios, leading to the formulation of the law of multiple proportions and his theory of atomism. In the early 19th century, British chemist John Dalton conducted experiments with gases and observed their behavior. It took nearly 2000 years for atomic theory to make a comeback. John Dalton and the law of multiple proportions Aristotle was considered the original social influencer and held significant influence in discrediting the concept of atoms for many years. However, Democritus's atomic theory faced opposition from Aristotle, who believed that everything on Earth was made up of four elements: earth, fire, Water, and air.

Although his theory faced skepticism and was later discredited, Democritus was on the right track regarding the idea that atomic composition influences substance properties. The Greek philosopher Democritus and his theory of atomosĪround 400 BC, a Greek philosopher named Democritus proposed a theory that everything in the world was composed of tiny, indestructible particles called "atomos," meaning "uncuttable." According to Democritus, the properties of materials were determined by the Type of atomos they were made of. But have you ever wondered what's inside a basketball? Similarly, people in generations past had a Curiosity about what lies within an atom. When You think of the word "basketball", the first thing that comes to mind is likely an orange sphere or your favorite athlete making an impressive dunk. The evolution of our understanding of atomic structure.Quantum mechanics and the Schrodinger- Wave equations.Niels Bohr's atomic model and the problem of stability.Ernest Rutherford and the gold-foil experiment.John Dalton and the law of multiple proportions.The Greek philosopher Democritus and his theory of atomos.Unveiling the Secrets of Atomic Models: Dalton, Rutherford, Bohr, and Heisenberg
